The Energy Drink are considered as non-alcoholic categorized drinks containing the nutritional information of caffeine, like guarana, glucuronolactone, taurine, ginseng, inositol, carnitine, B-vitamins, etc., which acts as stimulants. Within the past years, vivid energy beverages have been introduced in the Indian markets known as Energy Boost or supplements.
Several energy drinks contents with a high level of caffeine constituents which directly affects the nervous system of a human body. Such energy boost is rapidly and in larger aspects are marketed and advertised among the youngsters and others of the country and the manufacturers compare the effects of drinks likewise, the use of cocaine drugs.
We have a number of scientific research and reports which show the adverse consequences of excessive consumption of caffeine in mankind.
What are the Sources of Caffeine and Effects on Human Body?
The caffeine source is mostly rooted and found in tea, coffee[1], and other soft beverages. In energy drinks, caffeine added up to 80 mg per serving. And the energy drinks usually contents many types of water-soluble vitamins such as Niacin, Pantothenic acid, Vitamin B6 & Vitamin B12, and other substances such as amino acids, etc.
The high quantity of caffeine intake has a greater harmful impact on the health of an individual.
The conducted researches are the witnesses of several unwanted side effects of over 400 mg of caffeine intake per day; listed below are the side effects of caffeine.
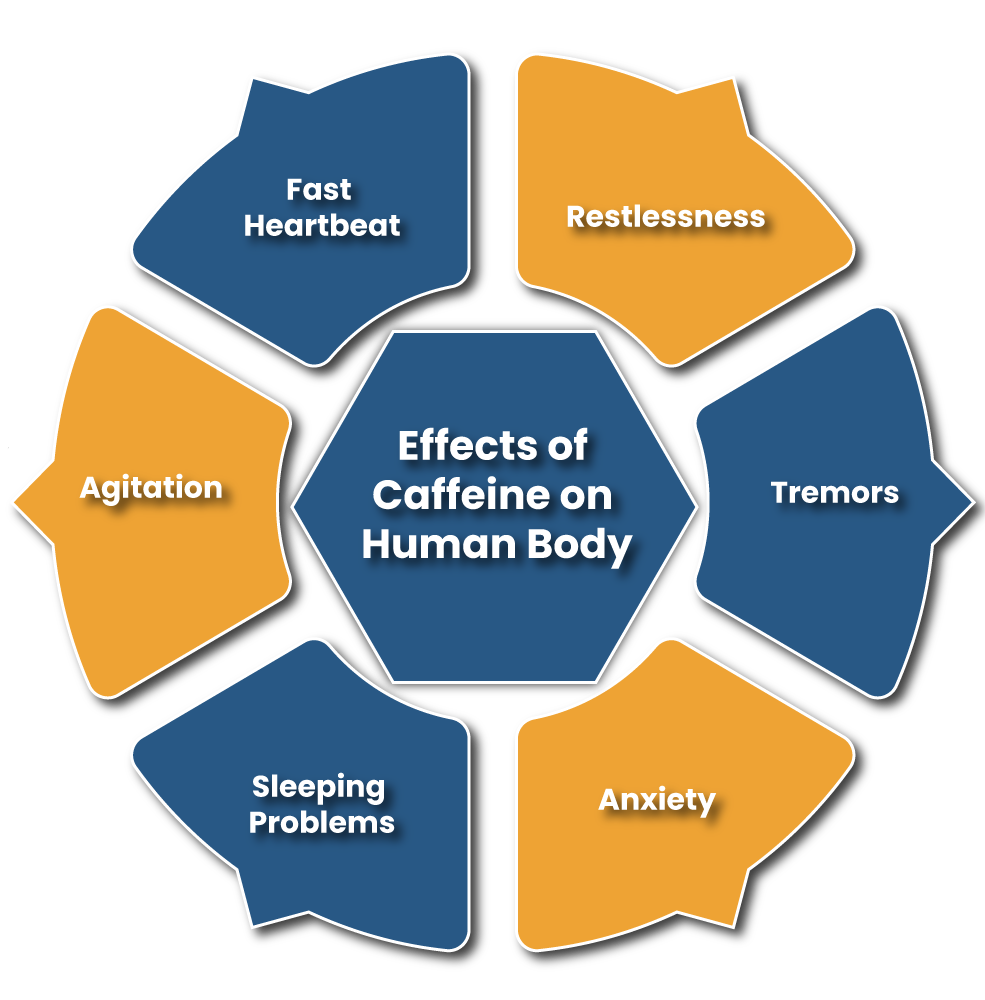

The aforesaid, side effects could be experienced by a person in case he wants to quit the habits of caffeine intake.
Read our article:Renew FSSAI License – When and How to Do it!
Standard Set By FSSAI for Energy Drink
- The FSSAI described the limit, and the number of Caffeine contents for the energy drinks beverages under the Standards of Caffeinated drinks. Classified the category of water-based flavored beverages, FSSAI defined the “so-called energy drinks” contains only high levels of nutrients and other ingredients.
- The Food Safety Standards defined the non-alcoholic beverages consisting more than 145 mg per liter of caffeine substance, must be leveled as “Caffeinated drinks”. The maximum permissible level of caffeine content in these beverages is 300 mg per liter regardless of the source.
- It is mandated to be labeled on such drinks about the consumable quantity of not more than 500 ml per day for an individual. This may denote the limits that how much energy drink, an individual should consume per day.
- The manufacturing company’s of Energy drinks must come with a marketing ideology of bold labeling regulations set by the appropriate authority in this context. They shall notify the “High Caffeine”, with the absolute quantity of caffeine in the product and must market such energy drinks with a display saying “not recommended for Minors, pregnant and lactating women, persons sensitive to caffeine’’ on the marketed products.
- The FSSAI prescribed a limit of a maximum of one ‘Recommended Daily Allowance’ level per day serving (i.e. 100% RDA) for the usage of Vitamins such as Vitamin B1, Vitamin B2, Vitamin B3, Vitamin B6, and Vitamin B12. The usage of amino acids such as Taurine, Inositol, etc. can also be done within their prescribed limits for per day serving.
It has now been a trend of rapid intake of energy drinks or beverages in the young section of the population. This factor has highly raised the concern and alertness of the Food and Safety Authority. The FSSAI always works on an agenda of Consumer Safety on its priority concern and is ensuring the implementations of stated rules and standards in regular enforcement activities.
The country India is one of the first among other countries to had codified rules & regulations for the sales of Energy Drinks. India drafted many conditions and necessarily required standards for Energy drinks manufacturers’ establishment engaged in manufacturing, marketing, or import of such drinks to meet the standards as one of the most difficult tasks in order to sell Energy drinks in the nation. This results in the involvement of taking assistance with the experts to comply and follow the FSSAI Regulations.
Prominent Cases of Energy Drink Which Underlined the Guidelines of FSSAI Standard
An Indian startup company Tzinga, manufactured by Hector Beverages Pvt. Ltd. claims to manufacture energy drinks at a lower rate than the other available energy drinks vide the lens of the Food Safety and Standards Authority of India. On dated 12th of May 2016, FSSAI ordered the immediate withdrawal of Tzinga products.
Why FSSAI Ordered For Immediate Withdrawal of Tzinga?
The FSSAI took samples and conducted a laboratory test of the drinks manufactured by the Tzinga Company. And the authority came with a strange report of finding the elements of ginseng and caffeine. Such manufactured products are capable of stimulating the nervous system which is a sign of highly unsafe for mankind’s consumption.
As per the FSSAI, Tzinga mislead and confused its customers about the benefits of energy drinks. The above-said company has also not heightened the strict and statutory warning labeled “High Caffeine Content” on its manufactured products. The manufactured drinks by the Tzinga establishment were highly unsafe for children, pregnant women, and for those who allergic to caffeine.
The authority has declared Tzinga drinks unsafe for consumption among the public at large because the manufactured drinks were prepared by excessive usage of caffeine and ginseng, which accordingly not be utilized to prepare as per the standards set by the FSSAI.
Current Status
After being banned for eleven months, The Company re-launched Tzinga in the Indian market, after receiving the approval from FSSAI. The ban was due to the presence of ginseng with caffeine, which is not allowed in an energy drink as per the standard set by the FSSAI.
Concluding Remark
Energy drinks usually circulate in Indian markets and are crucially presented as a booster for humans. It has been prevalent among the youngsters of the country to intake such drinks as a good sign of society civilization. There are lots of vivid energy drinks products launched in the Indian market, thus the concerned FSSAI authority set standards and difficult norms in selling such Energy drinks containing a high quantity of caffeine and other substances unsafe for the human body.
The authority limits in consumption of such drinks per day and to recognize and aware the innocent citizens about the “High caffeine Contents” product as the sole duty of the engaged establishment itself. Otherwise, for the betterment and public concern, FSSAI shall impose restrictions on selling likewise the beverage of Tzinga Company. Kindly associate with the Corpbiz expert to know more about the usage of caffeine prohibited under the energy drink by FSSAI Standards.
Read our article:Everything you need to know about FSSAI License Renewal & Validity











