Nearly every product that is sold in the European Union steak around a safety standard called CE. It is the short form of the term called “Conformité Européenne,” a french word. Manufacturers across the EU require to affix this logo on their product to ensure conformity against the EU’s directive and regulations. The CE mark is sort of manufacturer’s declaration that their product meets the required compliances against the applicable health, safety, and environmental norms.
It should be noted that any European organization does not own the CE mark. It is an autonomous standard with a certain set of rules and regulations that ensure the product’s safety on several testing grounds. In this blog, you will come across Documents required for CE certification in India.
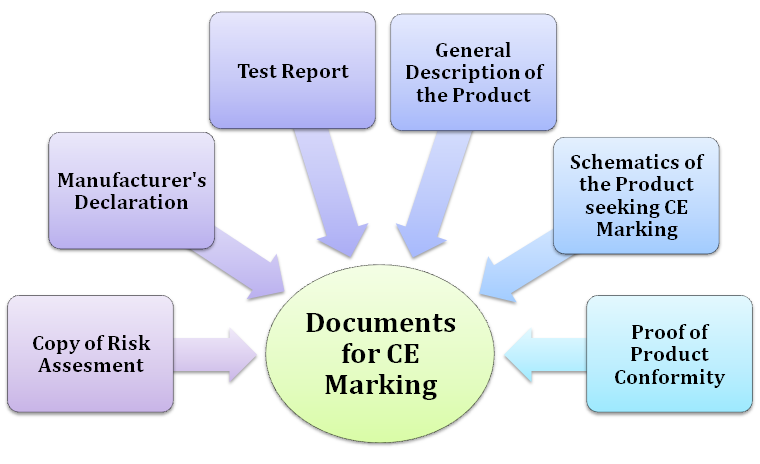

In general, CE mark can be raised by the manufacturer situated in the EU. However, it is not limited to the EU resident as the people from an overseas location can avail of this marking by appointing an authorized representative in the EU. Some directives guide the manufacturer to test its product by the certified third-party lab to ensure conformance with applicable standards.
These labs are also known as notified bodies in the EU. If the applicable directives discourage Notified Body’s use, then manufacturers can rely on their in-house testing facility for product conformity assessment. Each directive outline which conformity assessment procedures manufacture can undertake. Currently, you must take note that there are eight modules exist for the conformity assessment.
Read our article:All you need to know about the CE Certification Process
Technical Documentation Brief
The manufacturer must prepare the technical documentation, and it should enclose detail regarding risk assessment and adequate assessment. Technical documentation plays a prominent role in safeguarding an organization. It will retain the information about the evolution of the product and the evidence regarding its conformance with the required legislation. Technical documentation encloses a wide range of information about the product, including the test report. TD doesn’t follow any prescribed format as it could vary with regulations and directives.
Product Complies with the Essential Safety Requirements
All CE marking directive directs the manufacture to prepare technical documentation that declares that the product complies with the essential safety requirements. Technical documentation typically remains valid for ten years from the date on which the manufacturing of products was initiated. The updation of the technical documentation should be done accordingly as per changes in the product’s component, manufacturing process, or conformity assessment procedures. Now, let’s move to the main section i:e Documents required for CE certification in India.
Technical Document Required for the LVD, i.e. Low Voltage Directive
- Detail of the product’s design and manufacturing to assess conformity with the applicable legislation.
- Details description of the product for which CE marking is required.
- Schematic and drawing of the product’s component, circuits, and subassemblies along with the explanations for easy understanding.
- The functionality of the product.
- Detail of the existing standards governing the safety aspect of the product apart from the harmonized standards[1].
- Test report of the product generated by the notified body for the conformity assessment.
Technical Document Required for the EMCD, i.e. Electromagnetic Compatibility Directive (EMCD)
- Product identification mentioned in the technical documentation.
- A detailed description of the sample product.
- Evidence regarding the existing compliances governing the safety of the product. This should also cover the detailed procedure acquired by the applicant in order to meet the requirement for such complancies.
- Evidence regarding the European harmonized standards, if available.
- EMC assessment report.
- Declaration of the notified body in case if the manufacturer opted for procedure mentioned under Annex III of the EMCD,
- Test report
- Details of the examination carried out for the apparatus.
- Schematics of the testing
Notified Body Module
The notified body does the issuance of an EC type-examination certificate after testing the product as per the direction set out in module B to Decision 768/2008/EC. The NB’s keeps a copy of the certificate for future reference. Any changes made to the production process or the product must be intimated to the NB. EC type-examination enclose the following information.
- The name and complete address of the manufactures.
- Conclusion of the examination
- Applicable validity conditions.
EC-type Examination Certificate Issuance Criteria
- Its issuance of this certificate depends on the assessment of the tested model.
- The EC-type examination is performed by the notified bodies, which eventually decides whether the product is fit to use or not as per the EU’s directives. The NB is the autonomous body that adheres to no relationship with the manufacturers seeking certification.
- The manufacturer must streamline its production unit to set out conformity with the approved model seeking certification.
- The applicant should intimate NB about the manufacturing process changes without exception so that NB can conclude the report accordingly.
Conclusion
CE marking is absolutely mandatory for the manufacture who want sold their product in EU marketplace. Undoubtedly raising a declaration for conformity against the EU’s directive and regulations is an intimidating task for any manufactures. Thus; its important to cover all the basic points before diving into the conformity assessment of the product. It will help you overcome skepticism related to assessment formalities and standards.
The technical quandaries are apparent in obtaining any standard for the product. So in order to clear that hassle, make sure to feel free to connect Coprbiz expert today. You can also intimate our expert in case of any technical information regarding Documents required for CE certification in India.
Read our article:How to Get CE Marking Certification in India?










