Vaccine or drugs in India cannot be manufactured without permission from the government. There are tons of legal formalities that one needs to counter before involving drug manufacturing or distribution business. Currently, the existing drug producer in India works under the Drugs and Cosmetics Act, 1940. The Act encloses extensive guidelines that regulate the manufacturing and distribution of drugs in India. In this blog, we will look into the vast array of documents of Drug license.
Drug and Cosmetic Act, 1940
Drug and Cosmetic Act, 1940 was constituted by the government to set up legal guidelines against sales, manufacturing, and distribution of drugs and cosmetics in India. The Act has been evolving since its inception as it has undergone a couple of the amendments for the betterment of human life.

Classification of drug license
Depending on the definition of ‘Drug” in the Act, the drug license is categorized into five sections which are as follow:-
Manufacturing License
License grant to entities that deal with the manufacturing of drugs, including allopathic/ homeopathy medicines.
Sale License
The issuance of this license is done for selling drugs in the mainstream market. The sale license has been bifurcated into the following categories:
- Retail Drug License
- Wholesale Drug License
- Restricted Drugs
Loan License
Loan license is generally issued to firms who depend on outsourcing for producing the drugs.
Import License
This license category is a grant to those business entities which import the vaccination from overseas locations.
Multi-Drug License
The Multi-drug license is for the entities that own multiple pharmacies in different states under one brand name.
Read our article:A Complete Overview on Drug License Rules in India
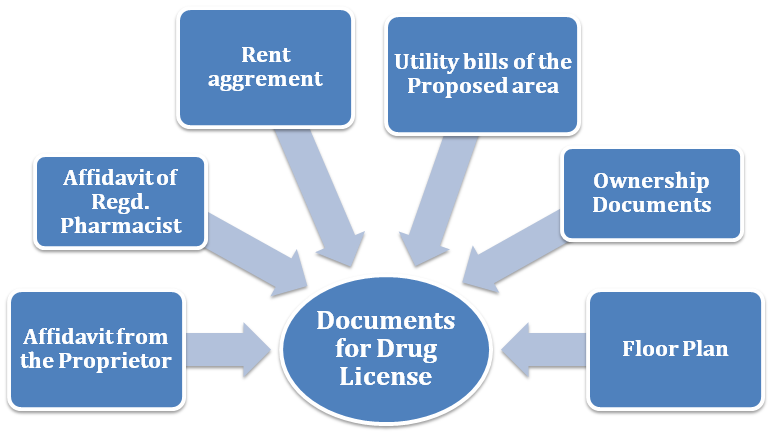
List of Documents of drug license
There are various and diverse documents needed for different types of drud License. Those are stated below in detail:-
Documentation for the sale license
- MOA, AOA, registration certificate, and address of the existing director in the case of Pvt. Ltd Company.
- Declaration in case of the proprietorship.
- Identification proof of the proprietor, partner, and director.
- Documents exhibiting the proof of the premises such as property’s ownership documents or lease agreement.
- Floor plan of the premises.
- Copy of the board resolution declaring that firm is ready to obtain a license.
- Documentation showing the availability of storage space.
- Copy of challan regarding depositing fee.
- Declaration on a stamp paper regarding the non-conviction of the proprietor.
- Declaration from the registered pharmacist.
Documents of Drug License for a pharmacist at a retail sale
- Aadhaar card
- Self-declaration (system generated)
- Academic and graduation proof of the applicant.
- Appointment letter.
- Copy of Experience certificate, if any
Documentation for a pharmacist at a wholesale sale
- Qualification proof
- Academic and graduation certificates
- Experience certificate
- Appointment letter
After gathering the above documents, users need to head over to the Food Safety and Drug administration’s online portal to fill up an online application. Once the applicant files the application with the legitimate information, the portal will prompt to upload the scanned copy of the above documentation. The portal further leads the applicant to the payment gateway to submit the application fee via digital payment modes.
As soon as the applicant completes the payment formalities, the portal will automatically render the option for generating challan. Upon receiving the application, the authority will initiate the scrutiny process for the same and directs the District Coordination Officer to inspect the business premises on the predetermined grounds. Once satisfies, the authority will eventually award the license to the applicant.
Critical points related to the Drug License in India
- The retailers and wholesalers need to display the drug license in the business premises expose to the general public.
- The licensee should not violate the provisions of the Drugs and Cosmetics Act, 1940, or else, he/she would be toil to pursue the business.
- Drug license holders are not allowed to sell medicines with low therapeutic value or harmful substances.
- Inexperienced candidates cannot be involved in distributing the legend drugs as it is a felony in the eye of the law.
- A candidate must have a minimum of 4 years of experience in drug distribution to starts a retail business of the vaccination.
- Any alteration in the workforce should be reported to the licensing authority within thirty days from the change date. Similarly, any change in the firm’s constitution should be reported to the licensing authority on a priority basis.
- The firm that fails to inform the authority about the amendment of the constitution shall be subjected to certain penalties mentioned under the law.
- The license would serve three-month validity periods in case of the amendment of the constitution provided the entity availed the new license in the meantime.
- Drug business lures tons of plenty in case of infringement of the bylaws mentioned in the Drugs and Cosmetics Act, 1940.
Conclusion
Drug manufacturers, distributors, wholesalers, retailers in India would be toil to pursue their business in the absence of the drug license. Any entities seeking involvement in drug sectors need to avail of the drug license through a legal process. The application for a drug license is available on the online portal of Food Safety and Drug Administration. As soon as you come across this application, you need to fill it with legitimate information and upload the requested documents. After completing this requirement, you will be prompted to complete the payment formalities regarding application fees.
Once done, the portal will transfer the application at the authority’s disposal, where relevant personals will examine it on the prescribed grounds. Feel free to connect CorpBiz’s experts in case if you need some professional-grade help in the direction of Documents of Drug License or other registration.
Read our article:How to Obtain Drug License in India?













